
Abstract
The composition and properties of boron nitride coatings are described. They can be used to coat and protect metallic and ceramic components used at elevated temperatures. For this purpose, the properties of boron nitride as well as the demands placed on starting materials in the production of such coatings are discussed. Additionally, examples for their practical application are given.
1 Introduction
Coatings are used to change the properties of substrate surfaces as required. The coatings described here protect metals and ceramics in a high-temperature environment and furthermore act as a release and lubricating agent. They inhibit or reduce the destruction of the substrates, e.g. by aggressive melts or gases.
The physical and chemical properties of coatings are mainly determined by the refractory fillers chosen. They can be tailor-made to different demands. It is possible to produce coatings showing good thermal conductivity or good thermal insulating properties. Electrically conductive or non-conductive coatings can be generated. In this way the production of superconductive coatings is possible. Other physical properties like friction or absorption of radiation can be controlled by coatings. Furthermore coatings may be used for marking purposes at high temperatures. Well known refractory fillers are for example Zr02, Y2O3, SiC and Si3N4.
In the following some coatings developed by the Büro für angewandte Mineralogie which contain hexagonal boron nitride (α-BN) as a refractory filler are looked at. The properties of boron nitride that are important for this application are discussed, and the composition and possible applications of such coatings are given. Only paintable coatings consisting of boron nitride suspended in a carrier liquid are considered here. These coatings may be applied similarly to paints by brushing, dipping or spraying. Other methods of application, which avoid carriers such as CVD and PVD, are not taken into account.
2 Properties of Boron Nitride
The hexagonal modification of boron nitride crystallizes in a layer structure similar to that of graphite (Fig. 1) [1]. A scanning electron micrograph shows plate-like particles of boron nitride powder used as a starting material for the production of boron nitride coatings (Fig. 2). The layers of boron nitride may be shifted easily so this substance may be used as a solid lubricant. The coefficient of friction for boron nitride compared with that of graphite is given in Fig. 3 in dependence on the temperature [2]. At temperatures up to 600°C, graphite shows a lower coefficient of friction than boron nitride. The strong increase of values between 600 and 800°C is caused by the oxidation of graphite. Here, boron nitride shows advantageous values. The climb in the curve of boron nitride between 800 ... 1000°C is caused by oxidation and the resulting formation of B2O3.
Boron nitride is stable in air up to 1000°C and in nitrogen atmosphere up to 2400°C. It shows good thermal conductivity combined with electrical insulation and good dielectrical properties [3].
Boron nitride shows chemical inertness and corrosion resistance to molten metals combined with very good nonwettability against many metallic and silicate melts, including Al, Mg, Cu and their alloys as well as glass and slags. The contact angle data for aluminium drops onto BN is compared with those of Al2O3 and Si3N4 in Fig. 4 in dependence on the temperature. Up to 900°C boron nitride is only poorly wettable by aluminium and shows a contact angle of 160°. Between 900 ... 1000°C the contact angle decreases, but is well above those of Al2O3 and Si3N4. Thus boron nitride is an ideal release agent for many shaping processes [4].
Fig 1. Structure of hexagonal boron nitride (α-BN) (left) and graphite (right)

Fig 2. Scanning electron micrograph (secondary electron image) of boron nitride powder
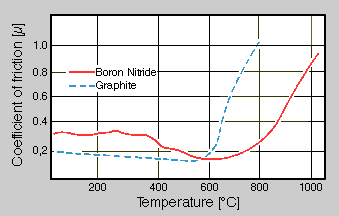
Fig 3. Comparison of friction data for boron nitride and graphite in air in dependence on the temperature
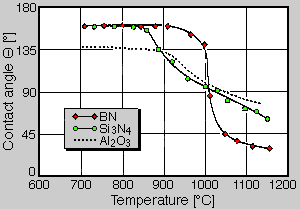
Fig 4. Contact angle data of aluminium on BN, Si3N4 and Al2O3 in dependence on the temperature
3 Composition of Coatings Based on Boron Nitride
Different types of boron nitride powders are available for the production of boron nitride coatings. They are suspended in a carrier liquid and often blended with a refractory binder.
3.1 Types of Boron Nitride
Boron nitride powders are commercially available with BN concentrations between 92 and 96 mass-% for refractory purposes and >98.5 mass-% for lubricating and shaping processes. B2O3 contents of up to 4 ... 8 mass-% are available, having carbon contents of up to 0.1 mass-% and metallic impurities up to <0.1 mass-%. Moreover, high-purity powders with BN concentrations of 99.5 ... 99.9 mass-% can be obtained for high purity melting purposes and electronic applications.
3.2 Carrier Liquids
The choice of a carrier liquid is mainly governed by technological aspects, but other points of view such as environmental and safety concerns must also be taken into account. Water is an inexpensive, readily available and non-hazardous option. However, forced drying of applied coatings will be necessary in most cases. Alternatively organic solvents such as alcohols (ethanol, 2-propanol) may be used, but these formulations have to be labelled according to hazardous materials regulations.
3.3 Refractory Binders
Boron nitride coatings can be produced without any refractory binder; the adherence of the particles can be achieved with their surface energy alone. Specific surface areas of at least 25 m²/g are necessary for this.
Most applications, however, require a coating with reasonable adherence which makes the use of refractory binders necessary. For coating ceramic substrates with low thermal expansion, phosphate-containing binders have proven to be well suited. These binders harden when heated to 800 ... 900°C and result in a strong yet brittle bond. They cannot be used for high-thermal-expansion substrates like metals. Here binders on the basis of Al2O3 for higher temperatures (>1000°C) or Al2O3·SiO2 for temperatures below 1000°C can be used. If the coating is meant to remain soft throughout the whole temperature range, clay minerals may be the binder of choice.
4 Application of Coatings Based on Boron Nitride
4.1 Range of Application
Metals as well as ceramics and graphite may be coated with boron nitride coatings resulting in good lubricating and release properties. In addition chemical reactions between melts and coated substrates are reduced or stopped and their life-time is increased.
Boron nitride coatings are used as a release agent in different shaping processes like hot pressing of ceramics, shaping of glass and coating of dies for metal casting. With good lubricating properties at high temperatures, coatings based on boron nitride are ideal release agents. For this reason they are also used for superplastic forming processes at temperatures up to 1000°C. Due to its high thermal stability, it has very often replaced the graphite and molybdenum disulfide used up until now.
The very good non-wettability property against melts is utilized in coating crucibles and combustion boats being a reasonable alternative to solid boron nitride. Other applications aim at the high thermal conductivity, e.g. coating and protecting thermocouple tubes before immersing them in melts.
4.2 Application
Boron nitride coatings should be applied on clean surfaces. Grit blasting may be necessary. Types of application include brushing, dipping and spraying. While brushing can be done with the coating as received, dilution of the coating with the particular carrier liquid may be necessary for spraying and dipping. Spraying of water-based boron nitride coatings on metals may be improved by heating the component to 100 ... 300°C. Only a thin layer (50 ... 100 µm) should be applied at a time in order to prevent cracking and peeling of the coating during subsequent drying. However thicker coatings may be achieved by applying several layers after drying between each application.
Boron nitride coatings should be dried thoroughly, water-based coatings preferably at temperatures of 105°C. Wet coatings should never be placed in service and never brought in contact with metal melts. After drying the coatings may be cured in order to harden the binder, however this depends on the type of binder used in the particular coating. This may occur when the coated component is placed in use for the first time.
It has been observed that the application of thin layers may be advantageous, especially when coating metals. As boron nitride has a low thermal expansion coefficient, stress may occur in thick coatings on high expansion substrates such as metals which may lead to chipping.
As boron nitride is a material with a hardness of 1 according to the Mohs scale, coatings containing boron nitride may be polished after drying with a soft cloth. Fig. 5 shows a scanning electron micrograph of a boron nitride coating, the right part having been polished. By this treatment the surface of the coating is smoothed and the pores are closed. However the softness of such coatings requires special attention to prevent damage by abrasion or sharp objects because damaged areas of the coating cannot protect the substrate sufficiently.

Fig. 5. Scanning electron micrograph (secondary electron image) of a boron nitride coating, partially smoothened by polishing
5 Conclusion
The present increased use of coatings is explained by the economical possibility of changing the properties of the surface of metallic and ceramic components. Advanced ceramic materials are increasingly used for this application. The composition of coatings based on boron nitride are disclosed and properties of boron nitride as well as corresponding coatings are discussed. At the same time references to the application and limitation of such coatings are made.
6 Literature
[1] A. Lipp., K. A. Schwetz, K. Hunold: Hexagonal Boron Nitride: Fabrication, Properties and Application. J. Eur. Ceram. Soc. 5 (1989) [1] 3-9.
[2] E. A. Smith: Graphite and boron nitride: aspects of structure, powder size, powder shape and purity. Powder Metallurgy 14 (1971) [27] 110-123.
[3] K. A. Schwetz, A. Lipp: Hexagonales Bornitrid. 1. Übersicht über Darstellung, Eigenschaften und Verwendung. Ber. Dt. Keram. Ges. 56 (1979) [1] 1-4.
[[4] M. G. Nicholas, D. A. Mortimer, L. A. Jones et al.: Some observations on the wetting and bonding of nitride ceramics. J. Mat. Sci. 25 (1990) 2679-2689.
Kurzfassung - Résumé - Resumen
Aufbau und Anwendung von Coatings auf der Basis von Bornitrid
Es werden Aufbau und Eigenschaften von Bornitrid-Coatings beschrieben. Sie eignen sich zur Beschichtung und zum Schutz von metallischen und keramischen Bauteilen, welche im Heißbereich eingesetzt werden. Dazu werden die Eigenschaften von Bornitrid sowie die Anforderungen an die Ausgangsstoffe zur Herstellung solcher Coatings diskutiert. Gleichzeitig werden Beispiele für ihren praktischen Einsatz aufgezeigt.
Composition de revêtements à base de nitrure de bore et applications
On décrit la composition et les propriétés de revêtements à base de nitrure de bore, qui peuvent être employés pour recouvrir et protéger des pièces céramiques utilisées à haute température. Dans cet esprit, on discute des propriétés du nitrure de bore et des exigences sur les produits de départ pour la préparation des dépôts. On donne par ailleurs des exemples d'application.
Composición y aplicación de revestimientos en base a nitruro de boro
Se describen la composición y las propiedades de revestimientos de nitruro de boro. Estos pueden ser utilizados para revestir y proteger componentes metálicos y cerámicos empleados a altas temperaturas. Para estos propósitos, se discuten las propiedades de nitruro de boro, asi como los requerimientos necesarios para las materias primas en la producción de tales revestimientos. Además, se presentan ejemplos de aplicaciones en la práctica.
from: Interceram 42 (1993) 5, p. 302-305, Verlag Schmid GmbH, Freiburg, Germany
![]() Download entire publication as pdf.
Download entire publication as pdf.
© 2023 Büro für angewandte Mineralogie · Dr. Stephan Rudolph · D-47918 Tönisvorst
These recommendations are believed to be correct. However, no guarantee of their accuracy is given. Therefore, purchasers shall make their own tests to determine suitability for their use. These products are offered for industrial and related uses (e.g. research and development) only. However the user must take the necessary precautions appropriate for products containing chemicals. This description does not imply the absence of any patents, the responsibility whatsoever solely rests with the user.

www.a-m.de