
Graphite can be used as refractory material at air up to 400°C (752°F), under inert gas however to 2500°C (4532°F). It shows good non-wetting behaviour against iron melts and many non-ferrous melts. Its crystalline structure is similar to that of boron nitride and finds application as a solid lubricant. Further graphite is used as an electrode material as well as for moulds and furnace linings. Special forms of graphite are colloidal graphites and carbon black (lamp black).

SEM-image (secondary electron image) of synthetic graphite in a magnification of 3000-times. The platelike particles are clearly visible, which are responsible for its lubrication properties
Colloidal graphites are colloidal solutions of graphite in water. By a colloidal solution one understands a solution, in which the solved particles are present in such a fine grain size, that a separation by filtration is not possible. In order to keep such solutions stable and to prevent precipitations, wetting agents and other additives are added. Colloidal graphites are mainly used for die-coatings, for example when casting brass in gravity die casting processes.
Carbon black is fine-grained carbon with specific surafces of up to 460m2/g. In die coatings soot is often blended with zircon and coke flour. Apart from that carbon black has an sliding effect when metals pass through dies in continuous castings. Important quantities of carbon black are used as fillers in the caoutchouc industry. Also it serves as a carbon supplier with the production of different metal carbides, for example those of silicon, titanium, vanadium and zircon. Carbon black is manufactured by an incomplete burning of carbon-containing gaseous, liquid or solid materials (gas soot, oil soot or flaming soot).
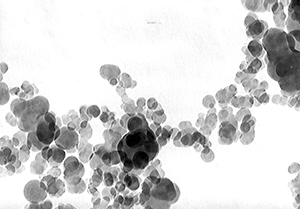
TEM-image (mag. 100.000-times) of furnace soot with a particle size of 56nm and a specific surface of 45m2/g
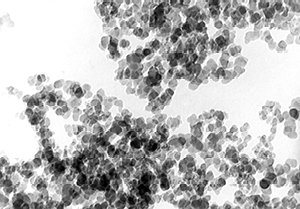
TEM-image (mag. 100.000-times) of gas soot with a particle size of 25nm and a specific surface of 100m2/g
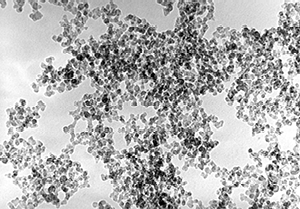
TEM-image (mag. 100.000-times) of gas soot with a particle size of 13nm and a specific surface of 460m2/g
.
Properties of graphite |
|
Formular |
C |
Density |
2.1 - 2.3 g/cm³ |
Hardness |
1 |
Melting point |
3800°C (6872°F) (sublimation) |
CAS-No. |
7782-42-5 |
EINECS-No. |
231-955-3 |
© 2023 Büro für angewandte Mineralogie · Dr. Stephan Rudolph · D-47918 Tönisvorst
These recommendations are believed to be correct. However, no guarantee of their accuracy is given. Therefore, purchasers shall make their own tests to determine suitability for their use. These products are offered for industrial and related uses (e.g. research and development) only. However the user must take the necessary precautions appropriate for products containing chemicals. This description does not imply the absence of any patents, the responsibility whatsoever solely rests with the user.
www.a-m.de